Corrosion control in closed cooling water systems
Key Highlights
- Closed cooling systems often use high-purity water, but oxygen ingress can lead to corrosion despite the absence of contaminants.
- Materials like carbon steel, copper alloys, and stainless steels are common, each with specific corrosion considerations and control methods.
- Corrosion mechanisms primarily involve electrochemical reactions, with oxygen playing a key role in metal oxidation and rust formation.
- Modern corrosion inhibitors such as nitrite, molybdate, and azoles are used to protect metal surfaces, with careful monitoring required to avoid over- or under-dosing.
- Additional control techniques include sidestream filtration and microbiological management using non-oxidizing biocides to maintain system cleanliness and integrity.
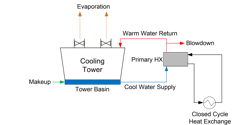
Countless chemicals, materials, consumer goods and other items are manufactured at industrial plants around the globe. Typically, these plants have numerous heat exchangers (HXs) for cooling process fluids. While some HXs may be directly served by large, once-through or open recirculating circuits — such as power plant steam surface condensers — many are on secondary, closed cooling water networks.
Common for many closed systems, and the focus in this article, is the use of high-purity water such as condensate for the fluid in the closed circuit. Often the question arises, “If pure water is the cooling medium, why is corrosion of concern? We will examine this issue next.
“Closed” systems are usually not completely closed
A precise definition of a closed system is that it “has no more than one point of interface with a compressible gas or surface... A system is considered ‘tight’ if make-up requirements do not exceed 5% of volume per year. Systems are often designed with a head tank for water makeup and to absorb volume changes per temperature and load fluctuations. This arrangement can allow oxygen to enter the cooling water, which, of course, influences the corrosion potential. Some closed systems may have a pressurized expansion or bladder tank to maintain constant water pressure. Makeup vessels or tanks are often located at the highest point in a closed loop to serve as an air release outlet for non-condensable gases that might otherwise accumulate in the system and can cause corrosion and pump cavitation.”1
Industrial and commercial closed cooling applications include:
- Pump bearings
- Lubricating oil
- Automated welders
- Metal casting coolers
- Steam turbine hydrogen coolers
- Chilled water systems
Typical closed system metallurgy
Carbon steel is the typical material for cooling water and condensate return piping, because of its high strength and economical cost. Copper alloys such as Admiralty brass and 90-10 copper-nickel are common for heat exchanger tubes due to their excellent heat transfer properties. The 304 and 316 stainless steels are also common for tubes or for plates in plate-and-frame HXs. Some heat exchangers have exotic materials for specialty applications, but our focus here is on the materials listed above.
Common corrosion mechanisms
Metallic corrosion is an electrochemical process, although some mechanisms such as erosion-corrosion are aided by mechanical influences. Consider the predominant corrosion process in most cooling systems, in which the cooling water is conditioned to a mildly basic pH.
Metal loss (“oxidation”) occurs at anodes, releasing ferrous ions (Fe2+) into solution. The free electrons flow through the metal to cathodes, where they react with dissolved oxygen at the surface to produce hydroxide ions (OH-). The basic reactions are:
Anode: Fe → Fe2+ + 2e-
Cathode: ½O2 + H2O + 2e- → 2OH-
Ion migration in the solution completes the electrical circuit. The overall reaction is:
Fe + ½O2 + H2O → Fe(OH)2
Further oxidation of Fe(OH)2 produces hydrated ferric oxide particulates (rust), which can cause additional problems.
The concept of anodes and cathodes is fundamental to corrosion understanding.
“In a closed system with low makeup, corrosion is often minimized because of oxygen depletion in the system. However, experience has shown that corrosion is not consistently reduced to acceptable levels because of oxygen depletion. This may be due to the fact that there are many potential paths for oxygen to enter the system and initiate corrosion. These path[ways] include oxygen saturated makeup water, the air-water interface in expansion tanks, and leaking pump seals.”1
Also, failures that have come from previous corrosion offer additional points for more oxygen ingress.
Copper is a more noble metal than iron, but in some environments such as water containing dissolved oxygen and aqueous ammonia, the oxygen will oxidize copper to Cu2+ which can then form a complex molecule with ammonia and dissolve. We will briefly return to this issue in the “Monitoring” section.
Corrosion control
The key point to remember is that corrosion inhibitors are designed “to protect the metal surface.”4 Some inhibitors form a protective layer at anodes and others at cathodes. This aspect is very important when establishing and monitoring chemistry programs.
In the middle of the last century and up until the early 1980s, a key ingredient in most cooling water chemistry programs for both open-recirculating and closed systems was sodium dichromate (Na2Cr2O7). The compound releases chromium ions that adsorb onto carbon steel to mimic a stainless-steel surface. Chemistry control was straightforward.
The realization in the 1980s that chromate treatment introduced toxic hexavalent chromium (Cr6+) to water systems led to a near total ban on chromate-based programs. The next section outlines the most common replacements. An important overall concept is that modern programs often include an additive(s) to maintain pH within a mildly alkaline range of say 8.5 to 10.5, as this minimizes general corrosion and “helps the corrosion inhibitor perform its function. [A common] buffer is sodium tetraborate.”1
Nitrite (NO2-)
Nitrite, often supplied as sodium nitrite (NaNO2) in granular form, is an anodic inhibitor. As shown by the following reactions, nitrite promotes the formation of a passive iron oxide layer on the metal surface.5
9Fe(OH)2 + NO2 → 3Fe3O4 + NH4 + 2OH + 6H2O
9Fe(OH)2 + NO2 → 3(Fe2O3) + NH4 + 2OH + 3H2O
Anodic inhibitors are known as “dangerous” inhibitors, for if the concentration is not or cannot be maintained within recommended guidelines, a small number of anodes can develop within a large cathodic field. Corrosion becomes intense and localized at the anodes, potentially leading to rapid through-wall pitting. A commonly recommended nitrite range is 500-1,000 ppm. However, when used with other inhibitors such as molybdate, lower nitrite residuals are possible.
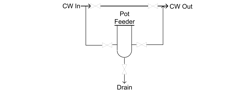
While water treatment chemical feed systems have become increasingly sophisticated over time, sodium nitrite feed does not have to be complex. With a tight system, it can be as simple as manually placing a charge of NaNO2 in a pot feeder and then injecting the chemical into the system via a slipstream. Once-per-week sampling may be sufficient to maintain residuals within recommended guidelines.
NO2- is a nutrient for nitrifying bacteria, which convert nitrite to nitrate (NO3-). The author once assisted with a project at an automobile assembly plant, where nitrifying bacteria were fouling the small, serpentine cooling water tubes in automated welders. A proposed solution was a changeover to molybdate treatment, whose basics are outlined in the next section.
In another case, this time at a power plant, the author and colleagues had to deal with a closed system where leakage was too great to maintain the recommended nitrite level for more than a day or so. Per the “dangerous” inhibitor discussion above, we suspended nitrite feed until the leaks were repaired, allowing establishment of a stable nitrite concentration.
Molybdate (MoO4-)
Sodium molybdate (Na2MoO4) has been utilized for corrosion control for decades, with additional interest arising when chromate chemistry was discontinued. Molybdate acts similarly to chromate and adsorbs onto the iron oxide matrix at anodes.
Fe2+ + MoO42- → FeMoO4↓
“This layer then may further evolve into what is known as a gamma iron complex. Research shows that molybdate acts as a pitting inhibitor per its ability to accumulate within the acidic part of a pit and block the corrosion process. A common control range for molybdate is roughly 1/3 of nitrite. Although molybdate is an oxyanion, it requires some residual oxygen to be effective. Enough dissolved oxygen may enter through the cooling water makeup or head tank to provide the needed amount of [O2].”6
Molybdate is an expensive chemical, so economics may influence its selection at plants with limited budgets. Nitrite-molybdate blends are a less expensive alternative. Molybdate is not a nutrient for microorganisms, but it is not toxic to microbes, either, and thus will not kill any organisms that might have entered the system.
Reducing agents
“Some programs utilize a reducing agent to remove dissolved oxygen and minimize the rate of corrosion of mild steel.”1 This treatment method was common for power plant feedwater systems, although significant changes have come to these programs in recent decades.7 Oxygen scavenger chemistry functions more effectively at higher temperatures, so it might not be applicable for some closed systems that operate at mild temperatures.
Copper alloy corrosion control
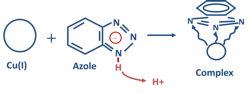
Azoles are common copper-alloy corrosion control agents in both open-recirculating networks and closed systems. Figure 5 shows the reaction between the most fundamental species, benzotriazole (BZT), and copper atoms on the metal surface.
The nitrogen atoms bond with copper, with the benzene rings forming a flat, protective layer on the metal surface.
Alternatives to BZT include tolyltriazole (TTA) and mercaptobenzothiazole (MBT). “Maintaining [azole] dosages in the range of 10 to 50 ppm will enhance effectiveness while remaining cost effective.”1 As a side note, other azoles that can better withstand degradation by oxidizing biocides than the above compounds have been synthesized for open-recirculating systems.
Additional water quality control issues
Additional techniques can be employed to help maintain closed cooling system cleanliness.
Sidestream filtration
Even in well-treated systems, corrosion products and particulates may accumulate over time. These solids can deposit on heat exchanger surfaces and influence heat transfer. Sidestream filtration is a straightforward method for particulate removal. Cartridge filters are one such design, where the filter pore size can be selected for optimum particulate removal.
Note: Sidestream filtration is a typical recommendation for open-recirculating systems where many particulates enter the circulating water via the makeup or especially from the air that passes through the cooling tower.
Microbiological control
Previously noted is that nitrite serves as a prime nutrient for nitrifying bacteria. Treatment with a traditional oxidizing biocide would deactivate nitrite and potentially damage system metals, but a non-oxidizing biocide, injected on a periodic basis, offers a potential control method. Common non-oxidizers include DBNPA (2,2-dibromo-3-nitrilopropionamide), isothiazolones and gluteraldehyde, among others. As with any chemical, safety when handling these materials and performing maintenance on feed systems is of the utmost importance. Anyone working with these chemicals should follow all safety data sheet (SDS) guidelines to the letter.
Monitoring
Both physical and chemical techniques are important for monitoring system performance. Fundamental is a water meter on the system makeup line to indicate if the network remains tight or if leaks have developed. A fixed meter is possible but from the author’s direct experience a portable ultrasonic meter with clamp-on sensors can provide accurate data, and can be easily moved to different locations.
Nitrite and molybdate concentrations can be readily analyzed on a UV-VIS (ultraviolet-visible) spectrophotometer. Common is once-per-week sampling with fresh chemical addition as needed. Beneficial analyses that can and should be added to this regimen include nitrate, ammonia, pH and specific conductivity. An increase in nitrate suggests the presence of nitrifying bacteria, while the appearance of ammonia indicates the additional presence of denitrifying bacteria. Such bacteria are important in many wastewater treatment processes, but their presence is unwelcome in closed cooling water circuits. Also, an increase in ammonia concentration suggests increased corrosion potential for copper alloys in a system. A drop in specific conductivity can also be an indicator of system leaks, with treated water being replaced by higher-purity makeup.
Recommended also is installation of a corrosion coupon rack in a bypass stream of the circuit.
Typical is to leave coupons in for one to three months or even longer, as with well-treated water the corrosion rate is very slow. The rate can be evaluated by taking before and after coupon weights. Pitting suggests a major chemistry control issue.
Glycol systems
Cooling systems subject to freezing conditions may have an ethylene or propolyene glycol solution as the coolant. Over time, and at higher temperatures, these glycols gradually break down to small-chain organic acids that accelerate carbon steel corrosion. Periodic withdrawal of degraded coolant and injection of fresh solution are necessary to minimize carbon steel corrosion. Acid generation of course lowers the pH, so regular pH monitoring is important for scheduling solution replenishment.
Conclusion
Closed cooling systems are integral equipment at many plants. While chemistry and microbiological control are usually much simpler than for open-recirculating or even once-through systems, careful attention is needed to minimize corrosion of piping and heat exchangers. Some failures can shut down an entire plant. Closed systems, like other plant equipment including remote cooling towers, are sometimes “out of sight, out of mind,” and receive insufficient attention until a failure occurs.
Disclaimer
This article offers general information and should not serve as a design specification. Every project has unique aspects that must be individually evaluated by experts from reputable water treatment and equipment firms. Also, any issues that could potentially have an environmental influence, for example, wastewater discharge from a proposed makeup, process or cooling water treatment system, must be presented to and approved by the proper environmental regulators during the project design phase.
References
1. K.A. Selby, “Closed Cooling and Heating Systems in Power Plants”; paper presented at the 22nd Annual Electric Utility Chemistry Workshop, May 7-9, 2002, Champaign, Illinois.
2. P. Robrege, Corrosion Basics: An Introduction, Third Edition; National Association of Corrosion Engineers (now the Association for Materials Protection and Performance), Houston, Texas, 2018.
3. Post, R., Buecker, B., and Shulder, S., “Power Plant Cooling Water Fundamentals”; pre-workshop seminar for the 37th Annual Electric Utility Chemistry Workshop, June 6-8, 2017, Champaign, Illinois.
4. This was a point of emphasis in the papers and seminars of the late Paul Puckorius, long-time cooling water expert and cornerstone member of the Cooling Technology Institute.
5. Information provided by Ray Post, (ret.), former cooling water program director for ChemTreat, Inc.
6. Water Treatment Essentials, ChemTreat, Inc., water treatment handbook, chapters of which are being periodically released on the ChemTreat website, www.chemtreat.com.
7. Buecker, B., “The importance of industrial water and steam treatment – Part 4”; Water Technology, December 18, 2023.
About the Author
Brad Buecker
Senior Technical Consultant with SAMCO Technologies
Brad Buecker currently serves as Senior Technical Consultant with SAMCO Technologies. He is also the owner of Buecker & Associates, LLC, which provides independent technical writing/marketing services.
Buecker has many years of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control, and results engineering positions with City Water, Light & Power (Springfield, Illinois) and Kansas City Power & Light Company's (now Evergy) La Cygne, Kansas, station. Additionally, his background includes eleven years with two engineering firms, Burns & McDonnell and Kiewit, and he spent two years as acting water/wastewater supervisor at a chemical plant. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 300 articles for various technical trade magazines, and he has written three books on power plant chemistry and air pollution control.
He is a member of the ACS, AIChE, AIST, ASME, AWT, CTI, and he is active with Power-Gen International, the Electric Utility & Cogeneration Chemistry Workshop, and the International Water Conference. He can be reached at [email protected] and [email protected].